|
Predict: Ionization energy (IE) is the energy required to remove an electron from an atom. Question: How does the radius of an atom affect the ability of the protons in the nucleus to hold on to and attract electrons? 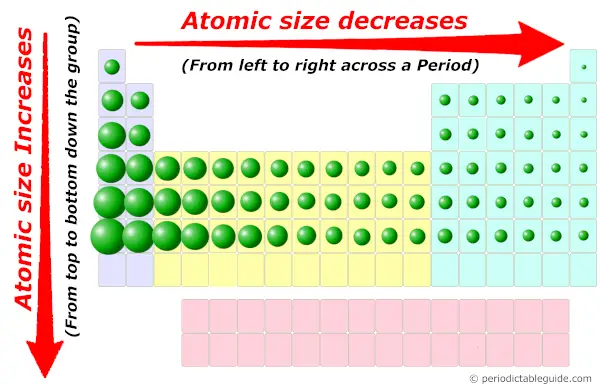
Gizmo PERIODIC TRENDS Answer Key – Activity B Why do you think the Cl-ion is larger than a neutral Cl atom?Ĭl is larger because it gained an electron and became stable Na is smaller because it has lost an electron and energy levelī. Why do you think the Na+ion is smaller than a neutral Na atom? Compare the radius of the neutral atom to that of the ion. Extend your thinking: The Gizmo enables you to examine ions, or atoms that have gained or lost electrons. How does your answer to the previous question explain the trend in radii across a period?Īs protons are added the attractive force increases, this If the proton number increases while the number of energy levels remains constant, what happens to the attractive force between the nucleus and the electrons?ĭ. What happens to the number of protons in the nucleus as you move from one element to the next across a period?Ĭ. As you move across a period, are new energy levels added?ī. Analyze: Consider how the number of protons might affect the size of the electron cloud.Īns: A. There is a stronger force of attraction pulling the electrons closer to the nucleus which makes a smaller atomic radius. Explore: Investigate other periods in the periodic table. Observe: What happens to the radius as you move across a period?Īns: we need to move the period across horizontally and see what happens to the radius, as we can see, moving the period from Cesium to Radon, not only the radius decrease, but also the atomic particles such as electrons, protons, and neutrons tend to increase (they are inversely proportional) Collect data: Beginning with Na, record the number of energy levels, number of protons, and atomic radius for each element in period 3 Predict: How do you think the radius of an atom will change as you move across a period (horizontal row)in the periodic table?Īns: The atomic radius decreases as you move across the period. Draw a conclusion: In general, what is the effect of the number of energy levels on the radius of an atom?Īns: As the group and number of energy levels increases, the atomic radius increases. What pattern do you see?Īns: The nucleus is getting bigger and the electron cells get smaller. Observe the radii of the elements in group 2. Observe: What happens to the radius as you move down group 1? As you do so, count the energy levels (shown as rings of electrons) in each atom. Collect data: Use the ruler to measure the atomic radii of the group 1 element. This is because the distance between a positively charged nucleus and a negatively charged electron increases as the number of energy levels increases. Predict: How do you think the radius of an atom will change as you move down a group (vertical column) in the periodic table?Īns: As you move down, the atomic radius will increase. Question: What factors affect the radius of an atom? Gizmo PERIODIC TRENDS Answer Key – Activity A Connect the right side of the ruler to the outermost electron, or valence electron. On the right side of the Gizmo, select Li. Hence, the atomic radius of hydrogen in this exercise is 53pm. Click “Save Radius” to record the value.Īns: By definition, the atomic radius is the measure of the space where the atom is found.  /chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
To measure the radius, drag one end of the ruler to the proton in the nucleus and the other end to the electron. To begin, check that H(hydrogen) is selected in Group 1 on the left. The atomic radius is a measure of the size of the electron cloud or the region where electrons can be found. Just as the thickness of a book changes… (Note: we are skipping the intro part so that we can directly jump into the Q&A section) The book thickness blocks the strength of a magnet’s ability to attract things. What is the relationship between the thickness of the book and the ability of the magnet to hold on to and attract paper clips?Īns: The thicker the book the less attraction the magnet will have with the paper clip.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
